|
About |
BD® offers a portfolio of products designed for your surgical needs.Visit booth #1511 to learn more about how we are partnering with you to advance your surgical goals and deliver quality patient care. |
Lorem ipsum dolor sit amet, consectetur
H3 Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed. Magna eget est lorem ipsum dolor sit amet.
Lorem ipsum dolor sit amet, consectetur
H3 Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed. Magna eget est lorem ipsum dolor sit amet.
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed.
- Lorem ipsum dolor sit amet,
- consectetur adipiscing elit, sed do eiusmod
- Habitant morbi tristique senectus et netus et
- Lorem ipsum dolor sit amet,
- consectetur adipiscing elit, sed do eiusmod
- Habitant morbi tristique senectus et netus et
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed.
- Lorem ipsum dolor sit amet,
- consectetur adipiscing elit, sed do eiusmod
- Habitant morbi tristique senectus et netus et
- Lorem ipsum dolor sit amet,
- consectetur adipiscing elit, sed do eiusmod
- Habitant morbi tristique senectus et netus et
H2 Lorem ipsum dolor sit amet, consectetur adipiscing
H3 Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed.
APIC Highlighted Solutions

BD® Surgiphor™ Antimicrobial Irrigation System
Surgiphor™ Antimicrobial Irrigation System is a pre-mixed, terminally sterile dilute povidone-iodine (PVP-I) surgical irrigation system. It is ready to use, enabling you to deliver a dilute PVP-I lavage to mechanically loosen and remove debris and foreign materials, including microorganisms, from wounds.
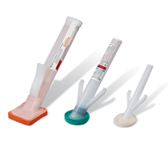
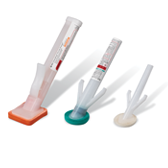
BD® ChloraPrep™ Patient Preoperative Skin Preparation with Sterile Solution
ChloraPrep™ patient preoperative skin preparation delivers standardized, powerful, persistent antimicrobial protection that is backed by more than 60 clinical studies and trusted by healthcare providers for more than 21 years.

BD® PurPrep™ Patient Preoperative Skin Preparation with Sterile Solution
BD PurPrep™ Patient Preoperative Skin Preparation with Sterile Solution is the first and only fully sterile povidone-iodine (PVP-I) plus isopropyl alcohol (IPA) in applicator form available in the United States. A sterile, simple and effective option when CHG plus alcohol is not ideal.
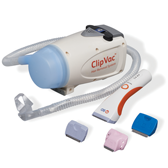
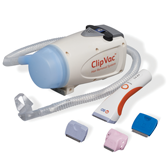
BD® Surgical Clippers and BD® ClipVac™ Presurgical Hair Removal System
BD Surgical Clippers and BD ClipVac™ Presurgical Hair Removal System work together to provide a comprehensive, one-step, preprocedure hair removal system that saves valuable patient-prep time.
The durable and ergonomic design of our clippers facilitates patient preoperative hair removal in a single pass and minimizes the risk of compromising the skin.
BD® Arista™ AH Absorbable Hemostatic Particles
Simple. Safe. Effective. Arista™ AH is a 100% plant based absorbable surgical hemostatic powder derived from purified plant starch. The power of Arista™ AH lies in its Microporous Polysaccharide Hemospheres, a patented blood clotting technology.
Let's have a conversation

H2 Lorem ipsum dolor sit amet, consectetur adipiscing
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit.
Bibendum est ultricies integer quis auctor elit.>>
Bibendum est ultricies integer quis auctor elit.>>
Bibendum est ultricies integer quis auctor elit.>>
Join BD® at our Satellite Symposia!
June 17, 2025
6–7:30 p.m.
West Ballroom 301A
Surgical site infections can lead to increases in hospital-reported costs, length of stay, and incidence of hospital-onset bacteremia and fungemia (HOB). Therefore, understanding the burden of surgical site infections and identifying risks in clinical practice are critical to improving patient outcomes and reducing patient morbidity and mortality. Join as we share and discuss alarming insights into operating room irrigation mixing practices and the clinical burden of surgical site infections and hospital-onset bacteremia and fungemia.
Speakers:
David J Weber, MD, MPH
Kalvin Yu, MD, FIDSA
Angela Vassallo, MPH, MS, CIC, FAPIC
Exhibitor Theater Presentation
June 18, 2025
9:15 – 9:45 a.m.
Every choice in the perioperative suite matters—where reducing complications and the risk of infections are critical to patient outcomes. Surgical site infections place your patients and hospital at risk: accounting for 1 of 5 HAIs, globally, increasing a patient’s post operative hospital stay by 7-11 days, and increasing costs to hospitals by $11,874 to $34,670 on average. Patients with an SSI have a 6-fold higher risk of developing hospital-onset bacteremia and fungemia (HOB). By applying evidence-based strategies, guidelines and care bundles, the risk of these outcomes may be reduced. Guidelines and bundles include a variety of interventions in the OR including glycemic control, parenteral antimicrobial prophylaxis, appropriate pre-operative hair removal, skin antisepsis, and surgical irrigation, but implementing these interventions may be inconsistent and difficult. This presentation will review the clinical and operational challenges faced by healthcare facilities in the operating room (OR) and explore strategies to create a comprehensive solution to address establish a new standard of care.
Speakers:
Madelyn Christie, RN, BSN
H3 Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed. Magna eget est lorem ipsum dolor sit amet.
H3 Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed. Magna eget est lorem ipsum dolor sit amet.
H2 Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod magus
Lorem ipsum dolor sit amet, consetetur sadipscing elitr, sed diam nonumy eirmod tempor invidunt ut labore et dolore magna aliquyam erat, sed diam voluptua. At vero eos et accusam et justo duo dolores et ea rebum.
Store Tab


360° rotation knob is effectively sized, contoured and located to enable easy instrument rotation.

360° rotation knob is effectively sized

360° rotation knob is effectively sized

360° rotation knob is effectively sized

H2 Lorem ipsum dolor sit amet, consectetur adipiscing
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit.
Bibendum est ultricies integer quis auctor elit.>>
Bibendum est ultricies integer quis auctor elit.>>
Bibendum est ultricies integer quis auctor elit.>>
H2 Lorem ipsum dolor sit amet, consectetur adipiscing
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit.
Bibendum est ultricies integer quis auctor elit.>>
Bibendum est ultricies integer quis auctor elit.>>
Bibendum est ultricies integer quis auctor elit.>>
H2 Lorem ipsum dolor sit amet, consectetur adipiscing
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit.
Bibendum est ultricies integer quis auctor elit.>>
Bibendum est ultricies integer quis auctor elit.>>
Bibendum est ultricies integer quis auctor elit.>>
H2 Lorem ipsum dolor sit amet, consectetur adipiscing
H3 Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed.
H3 Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed. Magna eget est lorem ipsum dolor sit amet.
H3 Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Habitant morbi tristique senectus et netus et malesuada fames. Bibendum est ultricies integer quis auctor elit sed. Magna eget est lorem ipsum dolor sit amet.
Visit us at booth #1511 to participate in the APIC '25 Knowledge Hunt!
Enter your information to speak with a BD® representative
Surgiphor™ Antimicrobial Irrigation System
Product description. Surgiphor™ Antimicrobial Irrigation System is an antimicrobial irrigation system containing 0.5% povidone iodine (PVP-I) in phosphate-buffered saline, potassium iodide and Vitamin E TPGS. PVP-I acts as a preservative to help inhibit microbial growth in the irrigation solution. Indication for use. Surgiphor™ Antimicrobial Irrigation System is intended to mechanically loosen and remove debris, and foreign materials, including microorganisms, from wounds. Contraindications: Surgiphor™ Antimicrobial Irrigation System should not be used in patients with known allergic reaction to any of the ingredients in the solutions. Surgiphor™ Antimicrobial Irrigation System should also not be combined with other irrigation or antiseptic solutions due to potential reactions and reduction in the effectiveness of the system. Not for use in neonates. Warnings. Do not use or mix with other cleansers, soaps, lotions, or ointments. Do not use for injection or infusion. Do not swallow. Do not use in eyes or ear canals. Discontinue use immediately if irritation or an allergic reaction occurs. Do not use if packaging is damaged or if seal integrity is compromised. Do not reuse Surgiphor™ solution after 24 hours. Precautions. Surgiphor™ solution may cause a temporary irritation and/or burning sensation on exposed skin in very rare cases. Surgiphor™ solution may cause allergic reactions such as rash or skin irritation in patients with iodine allergy. Anaphylaxis with the use of Surgiphor™ solution may occur in patients with severe iodine allergy. Federal law restricts this device to sale by or on the order of a licensed physician. Single patient use only. Not for at-home use.
ChloraPrep™ Patient Preoperative Skin Preparation with Sterile Solution
BD PurPrep™ Patient Preoperative Skin Preparation with Sterile Solution
Please consult product labels and inserts for any indications, contraindications, hazards, warnings, precautions and instructions for use. © 2024 BD. BD, the BD logo, the Sterile Solution logo, ChloraPrep and PurPrep are trademarks of Becton, Dickinson and Company. All other trademarks are the property of their respective owners. This document and the information contained herein is for general information purposes only and is not intended, and does not constitute, legal, reimbursement, business, or other advice. Furthermore, it does not constitute a representation or guarantee of cost-effectiveness, and it is not intended to increase or maximize payment by any payer. Nothing in this document should be construed as a guarantee by BD regarding cost-effectiveness, expenditure reduction, reimbursement or payment amounts, or that reimbursement or other payment will be received. The ultimate responsibility for determining cost-effectiveness and obtaining payment/reimbursement remains with the customer. This includes the responsibility for accuracy and veracity of all claims submitted to third-party payers. Also note that actual costs for products and services and any related expenditures vary, and that the information presented herein represents only one of many potential scenarios, based on the assumptions, variables, and data presented. In addition, the customer should note that laws, regulations, and coverage policies are complex and are updated frequently, and, therefore, the customer should check with its local carriers or intermediaries often and should consult with legal counsel or a financial or reimbursement specialist for any questions related to cost-effectiveness, expenditure reduction, billing, reimbursement or any related issue.
BD ClipVac™ Presurgical Hair Removal System
Intended Use: The intended use of the BD ClipVac System is to vacuum clipped hair and airborne contaminants generated from the clipping process. It should be used with the BD Surgical Clipper (REF 5513E). Only trained healthcare personnel should use BD ClipVac System and the BD Surgical Clippers. If the BD Surgical Clippers and BD ClipVac System are being used for the first time, see the training materials provided by BD. Operating Instructions: The bottom side of the clipper blade should remain flat and gently rest on the surface of the skin while clipping. To ensure hair is properly collected by the vacuum, never tilt the blade edge into the skin. For optimal performance, use on dry hair Disinfection Instructions: The BD ClipVac Vacuum Unit should be disinfected after each use, using one of the recommended disinfectant solutions listed below: Isopropyl Alcohol/Quaternary Ammonia solution Chlorinated bleach solution Inspection/Maintenance: The BD ClipVac Vacuum Unit requires no routine maintenance aside from cleaning system after each patient use. If a replacement is needed, contact your local BD representative or customer service. Service or attempted repair performed by unqualified personnel may result in a risk of injury, electric shock, fire, or permanent damage to the equipment and will void any warranty. Cautions: Use the BD ClipVac as described in the Operating Instruction. Avoid direct patient contact with the nozzle. Direct patient contact should be with the bottom side of the clipper blade. BD ClipVac Vacuum Unit, battery or charging adapter that appears to be defective, damaged, or not working. Do not cover vacuum unit to prevent obstructing air flow as this may cause overheating of vacuum unit. Do not re-use disposable filter assembly, it is intended to be single use only. Clean the vacuum unit after each patient use, refer to cleaning and disinfecting instructions. Do no submerse or spray the vacuum unit, battery, or charging adaptor with any liquid. Do not sterilize the vacuum unit, battery, or charging adapter. Do not short the battery terminals. Avoid excessive physical shock or vibration to the battery. Do not place the battery and charging adapter in a location where it is subjected to direct sunlight and keep away from other external heat sources. Do not dispose the battery with general waste. Never incinerate the lithium-ion battery. Do not replace charging adapter cord. Use only the battery and charging adapter supplied by BD. Do not attempt to modify or the vacuum unit, battery, or charging adapter as this can result in damage of the BD ClipVac system and will void the warranty. EMC Information of REF 5500: The BD ClipVac™ System is intended for use in the electromagnetic environment specified in the the Instruction For Use(IFU) provided with the BD ClipVac™. Warning: REF 5500E should not be used adjacent to or stacked with other equipment. If adjacent or stacked use is necessary, REF 5500E should be observed to verify normal operation in the configuration in which it will be used. REF 5500E has been tested and found to comply with the limits for the medical devices to the IEC 60601-1-2. These limits are designed to provide reasonable protection against harmful interference. This equipment generates uses and can radiate radio frequency energy and, if not installed or used in accordance with the instructions, may cause harmful interference to other devices in the vicinity. However, there is no guarantee that interference will not occur in a particular installation. If this equipment does cause harmful interference to the other devices which can be determined by turning the equipment off and on, the user is encouraged to try to correct the interference by one or more of the following measures; Reorient or relocate the receiving device. Increase the separation between the equipment. Connect the equipment into an outlet on a circuit different from that to which the other device(s) are connected. Consult the manufacturer or field service for help.
BD Surgical Clippers
Intended use: The BD Surgical Clipper REF 5513E is a rechargeable clipper used with charging adapter (REF 5514 series) and BD disposable blades only(REF 4406, 4403A or It is intended to remove head and body hair prior to any medical procedure requiring hair removal. The hair is removed by each blade oscillated by an electric motor. The BD 5513E Clipper will easily and effectively remove body hair and even the thickest hair from the chest with BD disposable blade REF 4406, scalp and other thick coarse hair with BD disposable blade REF and other difficult-to-clip areas of the body with BD disposable blade REF 4403A. The Clipper effectively removes wet or dry hair. Instructions for use: Only trained healthcare personnel should use the Surgical Clipper. If the Surgical Clipper is used for the first time, see the training materials provided by BD. Healthcare personnel should wear gloves when performing hair removal. Clipper should be disinfected prior to initial use. Suitable for use in healthcare facility environment. Check Clipper and blade for abnormal vibration or noise before patient use. See operating noise specifications listed in the Surgical Clipper specifications table in the Instruction For Use(IFU) provided with the BD Surgical Clipper. If abnormalities are found do not use the Clipper. Disinfection: Clipper should be disinfected after each use. Warning: BD blades(REF 4406, 4403A, and are single use only and specifically designed for use with the BD 5513E. The user assumes responsibility for appropriate use of this Clipper. Using blades not manufactured or approved by BD will void any warranty and patient results cannot be predicted. Re-use of blades may result in a nonfunctional product and could contribute to cross contamination; potentially putting patient safety at risk. The surgical clipper and charging adapter have been tested and found to comply with the limits for the medical devices to the IEC 60601-1-2. These limits are designed to provide reasonable protection against harmful interference. This equipment generates, uses and can radiate radio frequency energy and, if not installed and used in accordance with the instructions, may cause harmful interference to other devices in the vicinity. However, there is no guarantee that interference will not occur in a particular installation. If this equipment does cause harmful interference to the other devices which can be determined by turning the equipment off and on, the user is encouraged to try to correct the interference by one or more of the following measures; - Reorient or relocate the receiving device. - Increase the separation between the equipment. - Connect the equipment into an outlet on a circuit different from that to which the other device(s) are connected. - Consult the manufacturer or field service for help. EMC Information of REF 5513E/5514 series REF 5513E/5514 series needs special precautions regarding EMC and needs to be used according to the following EMC information in the Instruction For Use (IFU) provided with the BD Surgical Clipper. Warning: Use of REF 5513E/5514 series adjacent to or stacked with other equipment should be avoided because it could result in improper operation. If such use is necessary, REF 5513E/5514 series and the other equipment should be observed to verify that they are operating normally. Use of accessories, other than those specified or provided by the manufacturer of REF 5513E/5514 series could result in increased electromagnetic emissions or decreased electromagnetic immunity of REF 5513E/5514 series and result in improper operation. Portable RF communications equipment(including peripherals such as antenna cables and external antennas) should be used no closer than 30 cm(12 inches) to any part of REF 5513E/5514 series, including cables specified by the manufacturer. Otherwise, degradation of the performance of REF 5513E/5514 series could result. Caution: Prior to use: The clipper REF 5513E must be used with REF 5514 series charger. Disposable single use blades(REF 4406, 4403A and are designed for optimal use with 5513E only. Using blades not manufactured or approved by BD will void any warranty and patient results cannot be predicted. Keep charging adapter cord away from heated surfaces. Do not position charging adapter where it is difficult to unplug. Do not place the Clipper on the charging adapter until charging adapter is seated on a flat surface or securely mounted to a wall. Prior to charging Clipper ensure charging adapter is free of metallic debris. Do not expose to hot water, salt water, organic solvents or bleach solutions. Do not use with damaged blade, handle, or both. Do not take the housing apart as this can affect the watertight construction. Except when charging, it is recommended to unplug the charging adapter from the electrical outlet. This will reduce overall energy consumption and is compliant with best practices. Inspect treatment site for selection of appropriate blade. If skin irregularities are present, proceed with caution. Healthcare personnel should instruct patient to avoid sudden movement during clipping process. During use: Do not apply Clipper blade to injured skin area. Operating Clipper without blade could lead to injury. Do not use near flammable anesthetic, aerosol spray or oxygen — administering equipment other than nasal or mask types. Do not leave Clipper running without applying to skin for more than 1 minute as blade temperature may exceed 60 and potentially leading to thermal injury. Do not keep the Clipper blade applied to the same position of the patient’s skin for longer than 1 minute (these operations may result in blade surface becoming hot). In cases of minor injury, seek medical treatment if necessary.
Arista™ AH Absorbable Hemostatic Particles
Indications. Arista™ AH is indicated in surgical procedures (except neurological and ophthalmic) as an adjunctive hemostatic device to assist when control of capillary, venous, and arteriolar bleeding by pressure, ligature, and other conventional procedures are ineffective or impractical. Contraindications. Do not inject or place Arista™ AH into blood vessels as potential for embolization and death may exist. Warnings. Arista™ AH is not intended as a substitute for meticulous surgical technique and the proper application of ligatures or other conventional procedures for hemostasis. Once hemostasis is achieved, excess Arista™ AH should be removed from the site of application by irrigation and aspiration particularly when used in and around foramina of bone, areas of bony confine, the spinal cord, and/or the optic nerve and chiasm. Arista™ AH swells to its maximum volume immediately upon contact with blood or other fluids. Dry, white Arista™ AH should be removed. The possibility of the product interfering with normal function and/or causing compression necrosis of surrounding tissues due to swelling is reduced by removal of excess dry material. Safety and effectiveness of Arista™ AH have not been clinically evaluated in children and pregnant women. Because there have been reports of decreased amylase activity in newborns up to 10 months, absorption rates of Arista™ AH in this population may be longer than 48 hours. Arista™ AH should be used with caution in the presence of infection or in contaminated areas of the body. If signs of infection or abscess develop where Arista™ AH has been applied, re-operation may be necessary in order to allow drainage. Safety and effectiveness in neurosurgical and ophthalmic procedures has not been established. Arista™ AH should not be used for controlling post-partum bleeding or menorrhagia. Precautions. When Arista™ AH is used in conjunction with autologous blood salvage circuits, carefully follow instructions in the Administration section of the IFU regarding proper filtration and cell washing. Arista™ AH is intended to be used in a dry state. Contact with saline or antibiotic solutions prior to achieving hemostasis will result in loss of hemostatic potential. Arista™ AH is not recommended for the primary treatment of coagulation disorders. No testing has been performed on the use of Arista™ AH on bone surfaces to which prosthetic materials are to be attached with adhesives and is therefore not recommended. Arista™ AH is supplied as a sterile product and cannot be resterilized. Unused, open containers of Arista™ AH should be discarded. Do not apply more than 50g of Arista™ AH in diabetic patients as it has been calculated that amounts in excess of 50g could affect the glucose load. In urological procedures, Arista™ AH should not be left in the renal pelvis or ureters to eliminate the potential foci for calculus formation. Adverse reactions. None of the adverse events that occurred in a randomized prospective, concurrently controlled clinical trial were judged by the Data Safety Monitoring Board to be related to the use of Arista™ AH. The most common recorded adverse events were pain related to surgery, anemia, nausea, lab values out of normal range, arrhythmia, constipation, respiratory dysfunction and hypotension – all reported in greater than 10% of the Arista™ AH treated patients. The details of this clinical trial’s adverse events can be reviewed in the IFU supplied with the product and are also available at www.bd.com. Caution: Federal (USA) law restricts this device to sale by or on order of a licensed physician or properly licensed practitioner.
Please consult product insert for complete indications, contraindications, warnings, precautions, safety information and instructions for use.
BD is committed to keeping your personal data protected and secure. More information on how we protect your personal data can be found in our privacy statement and our cookie policy.